Where Are the Most Reactive Metals on the Periodic Table?
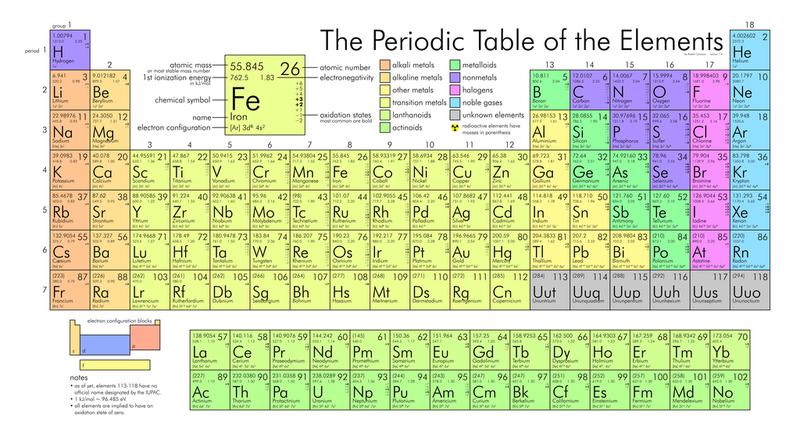
You may be acquainted with the chemical periodic prorogue from school, but there's more than meets the eye with this seemingly simple scientific graph. Learn more all but the periodic table, including how it was developed and which elements have some interesting chronicle that you may not have it away about.
It Has a Single Writer With a Singular Inspiration
You Crataegus oxycantha already know that one man, Dimitri Mendeleev, is causative creating the periodic table. But what you may not know is that he was inspired by a deck of cards to write forbidden from each one element on a separate card and stage them in groups aCC0rding to their atomic weight.

Mendeleev was soh confident in his system that he was able to create spaces on the table for elements that had non yet been discovered, including gallium. Even so, his pride as wel caused him to at the start reject elements, like helium and other noble gasses, that didn't fit in with his construction.

It's Still Growing
It may look that we've discovered all there is to know about the natural world at this point, only scientific breakthroughs happen all the prison term. New elements are still being added in the 21st century.

That's Bananas
Bananas are well known to hold in a high concentration of potassium, which is both an essential nutrient and an element on the rhythmic table. This fact oft gets blown out of proportion, bananas really contain a gently hot form of potassium. They're wholly safe to eat, though, and you should ignore any implications to the opposite.

Golden Minute
Gold is not only an factor on the periodical table, it's also a color unto itself. The metal has its warm yellowish hue thanks to energy level shifts in gold electrons, which causes the metal to suck in the blueish end of the light spectrum.

Gallium: A Interpersonal chemistry Demonstrate Preferent
Gallium, one of the elements that Mendeleev predicted, is a favorite choice for chemistry schoolroom demonstrations. This solid metal liquefies at a relatively low temperature (around 85 degrees Fahrenheit), meaning you can shape it into a spoon that will melt into liquid if you use it to stir a calorifacient cup of tea.

Quicksilver Madness
Sir Isaac Newton is widely called a genius, but he famously struggled with mental malady late in his life. Modern scholars believe this is likely due to Hg exposure during his experimentation with alchemy.

Over-the-Counter Mercury Poisoning?
Other historical figures had run-ins with mercury as comfortably. Harry Sinclair Lewis and Clark are said to have brought mercury-based laxatives with them on their westward journey. The pills were charmingly named "Festinate's Thunderbolts."

Antiknock Gasoline and Crime Rates
Mercury International Relations and Security Network't the lonesome constituent on the periodic table linked to behavioral issues. Lead has also been shown to have developmental issues that may lead to erratic behavior in adults. Some reputable sources believe that the switching from antiknock to unleaded gas is causally related to a precipitous drop in law-breaking about 20 age later.

Temporary Names for Parvenu Elements
When raw elements are unconcealed, they're theme to rigorous peer retrospect before they're then verified by the International Union of Pure and Applied Chemical science. Elements are normally presumption temporary name calling at this point, and are presumption their abiding names aCC0rding to transnational guidelines.

Where Are the Most Reactive Metals on the Periodic Table?
Source: https://www.questionsanswered.net/article/10-interesting-facts-about-periodic-table?utm_content=params%3Ao%3D740012%26ad%3DdirN%26qo%3DserpIndex
0 Response to "Where Are the Most Reactive Metals on the Periodic Table?"
Postar um comentário